Whisky wort fermentation produces ethanol, but also a variety of important flavours. Yeasts produce higher (fusel) alcohols and organic acids, which together form esters. Additionally ketones, sulphur compounds and phenols are formed. Whisky fermentation is quite similar to beer fermentation, but there are couple of important differences; the wort is not boiled, the distiller's yeast is usually propagated aerobically and the fermentations are usually not aeriated or temperature controlled (except the starting temperature). Unboiled wort allows the enzymes to continue their work and break down the oligosaccharides to increase the alcohol yield, but it also enables contamination with bacteriae and wild yeasts. If the yeast used is propagated aerobically, it is faster to start the fermentation and contains more sterols and fatty acids and thus the wort needs less oxidation or rousing.
 |
| Fermenting in Loch Lomond distillery |
Yeasts use simple sugars for their growth and energy metabolism. Simplified; when the yeast has oxygen, it produces water and CO₂ from glucose, but in anaerobic conditions it turns glucose into ethanol and CO₂ or alternatively glycerol. To reproduce, the yeast needs fatty acids, sterols and amino acids for its membranes and the organelles inside the cell. Oxygen is often needed in the production of these building materials.
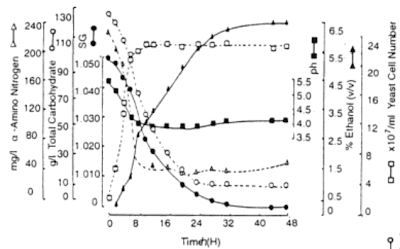
When yeast is pitched into the wort, it secures its energy reserves and if there are enough nutritients, it starts to reproduce by budding. The beefing up-phase is called the lag phase, and it is shorter if the yeast has been aerobically grown as the cells are usually full of nutritients already. The budding phase is called the log phase or the exponential phase, during which yeasts reproduce usually 3-4 times increasing the cell population about ten-fold. As the cells form new organelles and cell membranes, they produce a variety of different organic acids, fats and sterols including various intermediate products, some of which leak out of the cell into the wort. After that the nutritients and oxygen fall short and the cells do not reproduce, but try to produce sufficient energy to survive from the sugars, this is called the stationary phase. As the cells start to die or drop out from the fermentation, lactic acid bacteriae start to grow on the wort producing flavours typical of their metabolism, such as lactic acid and several lactones.
The amount of higher alcohols depends on the yeast growth; basically the more the yeast grows, the more higher alcohols are formed. Therefore aeriation of the wort, high nitrogen, and high temperature promote fusel alcohol production. Ale strains usually produce more fusel alcohols than lager strains, partly because of the higher fermentation temperatures. Fusel alcohols themselves are not a desired flavour in the wort - producing usually a sharp, solventy notes - but together with acids they form esters, which are important and desired flavour compounds in whisky as they produce various fruity and flowery notes.
| Amino acid | Fusel alcohol |
| Leucine | Isoamyl alcohol |
| Valine | Isobutanol |
| Isoleucine | Active amyl alcohol |
| Phenyl | 2-phenylethanol |
| Tyrosine | p-hydroxyphenylethanol / tyrosol |
| Tryptophan | Tryptophol |
| Methionine | Methionol |
Table1. Aminoacids metabolise into different fusel alcohols
 Ester formation depends on the amount of fusel alcohols and organic acids in the wort, but also on the activity of alcohol acetyltranferase enzymes (ATAase I and II), which in turn depends greatly on the yeast strain. Esters in the fermentation can be classified into two groups: The acetate esters (acetate+alcohol) and the ethyl esters (ethanol+fatty acid). The acetate esters are usually formed in greater amounts, but the ethyl esters can be very aromatic even in low concentrations. Common descriptors for the aromas of esters are listed in the table below. The short chain fatty acid esters (C6, C8) are formed early in the fermentation, the medium chain esters (C10,C12) quite evenly throughout the fermentation and the longer chain esters (C16) mostly at the cell-death phase. Increased cell growth usually results in lower levels of esters, due to lower levels of free fatty acids in the wort, as fats are used to build cell membranes. Organic acids are formed throughout the fermentation and at high levels they produce notes of vinegar, vomit and barnyard. The right proportion of fusel alcohols and free fatty acids or acetate is crucial when producing estery wort and avoiding the solventy off-notes from the excess alcohols and on the other hand the rancid aromas from the excess free fatty acids. An estery, fruity wort can be produced with warm long fermentations, high original gravities, high pitching rates with aerobically grown yeast and low nitrogen barley. Increased glucose levels tend to produce more short chain esters, for example isoamyl acetate with a typical banana aroma. High fermentation temperatures usually produce more acetate esters with mainly fruity aromas, but also medium-long chain ethyl esters, which can give an oily and waxy texture to the flavour.
Ester formation depends on the amount of fusel alcohols and organic acids in the wort, but also on the activity of alcohol acetyltranferase enzymes (ATAase I and II), which in turn depends greatly on the yeast strain. Esters in the fermentation can be classified into two groups: The acetate esters (acetate+alcohol) and the ethyl esters (ethanol+fatty acid). The acetate esters are usually formed in greater amounts, but the ethyl esters can be very aromatic even in low concentrations. Common descriptors for the aromas of esters are listed in the table below. The short chain fatty acid esters (C6, C8) are formed early in the fermentation, the medium chain esters (C10,C12) quite evenly throughout the fermentation and the longer chain esters (C16) mostly at the cell-death phase. Increased cell growth usually results in lower levels of esters, due to lower levels of free fatty acids in the wort, as fats are used to build cell membranes. Organic acids are formed throughout the fermentation and at high levels they produce notes of vinegar, vomit and barnyard. The right proportion of fusel alcohols and free fatty acids or acetate is crucial when producing estery wort and avoiding the solventy off-notes from the excess alcohols and on the other hand the rancid aromas from the excess free fatty acids. An estery, fruity wort can be produced with warm long fermentations, high original gravities, high pitching rates with aerobically grown yeast and low nitrogen barley. Increased glucose levels tend to produce more short chain esters, for example isoamyl acetate with a typical banana aroma. High fermentation temperatures usually produce more acetate esters with mainly fruity aromas, but also medium-long chain ethyl esters, which can give an oily and waxy texture to the flavour.
Ester Name
|
Odor or occurrence
|
Allyl hexanoate
|
pineapple
|
Benzyl acetate
|
pear, strawberry, jasmine
|
Bornyl acetate
|
pine
|
Butyl butyrate
|
pineapple
|
Ethyl acetate
|
nail polish remover, model paint, model airplane glue
|
Ethyl butyrate
|
banana, pineapple, strawberry
|
Ethyl hexanoate
|
pineapple, waxy-green banana
|
Ethyl cinnamate
|
cinnamon
|
Ethyl formate
|
lemon, rum, strawberry
|
Ethyl heptanoate
|
apricot, cherry, grape, raspberry
|
Ethyl isovalerate
|
apple
|
Ethyl lactate
|
butter, cream
|
Ethyl nonanoate
|
grape
|
Ethyl pentanoate
|
apple
|
Geranyl acetate
|
geranium
|
Geranyl butyrate
|
cherry
|
Geranyl pentanoate
|
apple
|
Isobutyl acetate
|
cherry, raspberry, strawberry
|
Isobutyl formate
|
raspberry
|
Isoamyl acetate
|
pear, banana (flavoring in Pear drops)
|
Isopropyl acetate
|
fruity
|
Linalyl acetate
|
lavender, sage
|
Linalyl butyrate
|
peach
|
Linalyl formate
|
apple, peach
|
Methyl acetate
|
glue
|
Methyl anthranilate
|
grape, jasmine
|
Methyl benzoate
|
fruity, ylang ylang, feijoa
|
Methyl butyrate (methyl butanoate)
|
pineapple, apple, strawberry
|
Methyl cinnamate
|
strawberry
|
Methyl pentanoate (methyl valerate)
|
flowery
|
Methyl phenylacetate
|
honey
|
Methyl salicylate (oil of wintergreen)
|
Modern root beer, wintergreen
|
Nonyl caprylate
|
orange
|
Octyl acetate
|
fruity-orange
|
Octyl butyrate
|
parsnip
|
Amyl acetate (pentyl acetate)
|
apple, banana
|
Pentyl butyrate (amyl butyrate)
|
apricot, pear, pineapple
|
Pentyl hexanoate (amyl caproate)
|
apple, pineapple
|
Pentyl pentanoate (amyl valerate)
|
apple
|
Propyl acetate
|
pear
|
Propyl hexanoate
|
blackberry, pineapple, cheese, wine
|
Propyl isobutyrate
|
rum
|
Terpenyl butyrate
|
cherry
|
Table2. Common esters and their aromas.
Diacetyl is an important flavour compound producing slick, buttery mouthfeel from concentrations of about 1ppm and at higher concentrations butterscotch or even cheesy flavours, and is usually considered as an off-note. It arises from the nitrogen metabolism during the exponential phase as the cells convert aminonoacids into ketones (such as diacetyl) and back to different aminoacids, but in the late stationary and the cell-death phases the cells use ketones in their metabolism as the sugars are running low. Brewers and distillers usually allow a diacetyl-rest period after the active fermentation to clear the wort of excess ketones. Too short fermentation time usually results in excess diacetyl. Heating, for example during distilling, increases the formation of diacetyl from other ketones. Diacetyl is quite volatile with a boiling point of 88⁰C and very hard to remove from the spirit even with column distillation.
 |
| Sulphur mining in an active volcano, Java (from zmescience.com) |
Yeast metabolism produces many sulphur compounds, mostly sulphur dioxide (SO₂, burnt matches). SO₂ is easily reduced to hydrogen sulphide (H₂S, rotten eggs), which is very volatile and easily carried out of the wort if sufficient CO₂ is formed. Slow fermentations due to for example low temperature, low pitching rate, contamination or unhealthy yeast could fail to produce enough CO₂, which leads to high levels of H₂S in the wort. Some highly aromatic sulphur compounds such as dimethylsulphide (DMS) and -trisulphide (DMTS), dimethylsulphoxide (DMSO), S-methyl methionine (SMM), dithiapenthyls (DTPOH, DTPA) and various mercaptans originate mostly from the malt, but are metabolised by yeast and their concentrations can be either elevated or decreased during fermentation. Methione and cysteine are amino acids with a sulphur chain, which can be broken down during cell growth and energy metabolism. Starved cells can also turn into catabolic state (autophagosytosis), in which they break down their cell organnelles (and amino acids in the process) to produce energy, and this produces excess sulphur. This is probably why anaerobically grown brewer's yeast together with distiller's yeast produces more sulphur compounds than either one used alone. Starved brewer's yeast (cropped from the brewery, not from an aerobic propagation or a lab) produces over twice as much aromatic sulphur compounds than fresh yeast of the same strain. Distiller's yeast used alone produces slightly less aromatic sulphur than a common ale yeast, probably because of its better nutritional state. The aromatic sulphur compounds are not necessarily off-notes, but are in fact needed for full-bodied and complex aromas (in the right proportions, of course).
Phenols in whisky are mostly derived from peat burnt in the maltings, but some very flavour-active phenol compounds can be produced by yeasts. Wild yeasts produce significant amounts of 4-vinyl guaiacol, which has a very potent phenolic aroma. Phenolic note has been considered an off-note in brewing and therefore the brewers have usually chosen strains that do not have a functioning gene for 4-vinyl guaiacol-production, exceptions include most hefeweisen and rauchbier yeasts and of course the lambics brewed with wild yeasts. Apparently also the commercial distiller's yeasts are lacking the "phenolic off-flavour" genes.
The picture below sums the simple reactions involved in the flavour formation during alcoholic fermentation.
The picture below sums the simple reactions involved in the flavour formation during alcoholic fermentation.
 |
| Flavour formation from alcoholic fermentations. (Ramsay 1982) |
Bryce JH et al (ed). Distilled spirits: Production, technology and innovation. Nottingham Univ Press 2008
Piggott JR et al (ed). The science and technology of whiskies. Longman 1989
Querol A, Fleet GH (ed). The Yeast Handbook. Springer-Verlag Berlin 2006
Russell I (ed). Whisky, technology, production and marketing. Academic Press 2003
Walker GM, Hughes PS (ed). Distilled spirits, new horizons: energy, environment and enlightenment. Nottingham Univ Press, 2010
White C, Zainasheff J. Yeast. Brewers Association 2010
"As the cells start to die or drop out from the fermentation, lactic acid bacteriae start to grow on the wort producing flavours typical of their metabolism, such as lactic acid and several lactones."
ReplyDeleteIs this favorable in scotch production?
It depends on whether the distiller wants the LAB-effect (usually sweet, sometimes estery or smoky, depending on the lactic aced bacteriae colonisation in the distillery). About half of the distillers use short fermentation times (with practically no LAB-effect) and about a quarter use very long fermentation times (over 70h) which probably lead to greater bacterial contamination of wort. I will put up a table on different practices as soon as I have some spare time.
DeleteThanks Teemu. Appreciate your insight and knowledge.
DeleteAnother great article. thanks.
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteGreat article, however I've noted a couple minor nits.
ReplyDelete1.Yeast do not make methanol. Some true anaerobes can produce methanol, but not in wash fermentation conditions. Methanol in distilled beverage production is almost entirely from terminal methyl groups of pectins and the levels of these is very low in grains, however can be substantial in pectin rich fruits (for brandies).
2. Paragraph 2 ignores the Crabtree effect. It's actually difficult to get aerobic yeast metabolism (respiration)from Saccharomyces. This needs to be distinguished from growth in an oxygen rich environment.
3. FAs are used to make cell MEMBRANES and never cell WALLs. Cell walls consist of mannose-protein complex an glucans.
3. LAB produce hydroxy-fatty acids, then yeast may convert these into gamma-lactones (note however that "whisky lactone" is from the barrel). LAB don't wait for yeast to finish, however their metabolic activity is relatively slow.
4. Fatty acids of increasing length (via liposynthesis metabolism) are produced intracellularly during yeast growth. However when some growth limit is reached, yeast cease to elongate the FAs. Any short/mid length FAs are dangerous as they have a detergent effect on the lipid bilayer membranes. Yeast instead esterify these FAs, at an energetic cost, as you indicate.
5. Ethyl esters are the most abundant, with ethyl-acetate (ester of ethanol) dominating others by an order of magnitude or more, and is often the dominant odor of white-dog.
6. Oak barrels are very a substantial source of phenolics in whisk[e]y, including vanillin, cinnamic acid and guiaiacol.
sincere thanks for the great blog ;^)
Thanks for the corrections and revisions. As you can see, I am no biochemist. I admit making some crude simplifications in exchange for readability.
DeleteThank you for your article , I am interested in trying to find an average or ideal time in terms of hours that would be require to ferment 2000L , that will later be distilled , is there heat involved other than the chemical process
DeleteThanks for a fine article. I assume lactic acid bacteriae must originate in some contaminated part of the distiller's system. They don't just appear, can they be added? Should a contamination be started?
ReplyDeleteIn a brewery (typically closed-fermentation) the lactic contamination would probably come from improper cleaning procedures or an oddity in the mashing process. Distilleries quiet often ferment open-air so wild yeast bacteria, etc have access. Also, the wash is acidified to create a positive environment for the yeast but it creates a primed environment for the lacto
Deleteediting^
Deletenormally lacto is not added to a mash/wash but are normally present to some extent anyway. Its somewhat common to add brettanomyces for beer tho
Great article. Lots of details information of whisky making!
ReplyDeletethanks very much for your sharing and this website opens a great door for me.
Are there any distilleries boiling their wort/wash before fermentation? Aren't their alternative routes to lacto accumulation in the wash such as mixed cultures? Thanks for the great blog. I'm coming at this from a brewer's perspective, so forgive my lack of insight on the topic. I'm sure there are economic, as well as tried and true reasons behind whisky fermentations and why they have not changed much over the last 100 years.
ReplyDeleteNot any Scottish distilleries that I know of boil their mash. Deliberate addition of lactobacilli in the form of mixed cultures is actually illegal. Most lactobacilli come probably from the mash tuns.
ReplyDeleteI'm curious how these breweries opening distilleries (e.g. Adnam's, Eden Mill) are making whisky without boiling their mash and potentially spreading various bacteria from the malt into their production facility. I'm new-ish to whisky but can see that there's an obvious sensory benefit from the LAB effect. However, I'm still trying to wrap my head around if there is any sort of control in regards to the amount of LAB present during fermentation in order to avoid ethanol losses at the still. It seems that the accumulation of Lactobacilli in washbacks would yield inconsistent fermentations. Your blog has been a lot of help, thanks again.
ReplyDeleteI guess it is just a matter of cleaning, cleaning, cleaning. As any serious brewer already knows. Consistent LAB does give quite consistent fermentations, as in lambic. Not factorylager-like, but consistent enough. For gueuze blending at least ;)
DeleteWhat effect does the boiling of the yeast itself in the distillation pot have on flavor of the distillate? Is it desirable to drop the yeast out of the fermented wort to minimize the amount of yeast cells in the distillation boiler?
ReplyDeleteYeast cells in distillation probably provide more fats and sulphur. Cognac distilled "on the lees" is said to be more fruity (probably more esters from fats+alcohols). Should be quite easy to burn the yeast on the bottom of the still and add sulphury vegetable off-notes.
DeleteHi, great read! Can you advise which article or book you got the figure: Flavour formation from alcoholic fermentations. (Ramsay 1982) from?
ReplyDeleteThanks
Current developments in malting, brewing and distilling: Proceedings of the Aviemore Conference held at the Aviemore Centre, Aviemore, Inverness-shire, 24-28 May 1982 Paperback – 1 Jan 1983
DeletePublisher: Institute of Brewing (1983)
ISBN-10: 0900489057
Thanks for a great article. I would like to know how much fusel oils are removed in pot still when compared to modern distillation column.
ReplyDeleteGreat article, I would like to know what is the optimum temperature of fermentation, if the temperature goes up 30 centigrade what will happen to the resultant spirit
ReplyDeleteI think, too much esters is not good for whisky.
ReplyDelete